
Innovations in Science
Grants at a Glance
MDA’s research program awards grants to the world’s best scientists investigating promising theories and therapies that may accelerate treatments and cures for families living with muscular dystrophy, ALS and related neuromuscular diseases.
Grant - Summer 2011 - DMD/BMD — Michael Rudnicki, Ph.D.

MDA has awarded a grant totaling $375,000 over three years to Michael Rudnicki, director of the Regenerative Medicine Program at Ottawa Hospital Research Institute and a professor in the department of medicine at the University of Ottawa, Canada. This funding will support Rudnicki's continuing studies on the function of muscle satellite cells in mice with a disease resembling human Duchenne muscular dystrophy (DMD).
DMD is caused by any of a number of mutations in the gene for the muscle protein dystrophin that result in a complete absence of the dystrophin protein in the skeletal muscles and heart. The closely related disease Becker muscular dystrophy (BMD) is due to mutations in the dystrophin gene that result in dystrophin protein that's only partially functional.
Muscle satellite cells are required for the growth and repair of skeletal muscles. In previous studies, Rudnicki and his colleagues have found that there is a subset of muscle satellite cells that function as "satellite stem cells," giving rise to the satellite cells that then become muscle.
They've also found that satellite stem cells do not function properly in dystrophin-deficient mice; and that a protein called WNT7a stimulates proliferation of satellite stem cells and benefits normal and dystrophic muscle.
In this new set of experiments, Rudnicki's team will investigate the nature of the defect in satellite stem cells in dystrophin-deficient mice and the mechanism through which WNT7a treatment increases the numbers of these cells.
The researchers also will investigate the possibility of using WNT7a as a treatment for DMD or BMD, through experiments in dystrophin-deficient mice.
Rudnicki, who has received MDA support for several years, says, "MDA funding has been critical in allowing my lab to investigate the fundamental mechanisms that regulate the function of muscle stem cells."
Funding for this MDA grant began August 1, 2011.
Grantee: DMD/BMD — Michael Rudnicki, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - DMD/BMD — Josephine Nalbantoglu

Josephine Nalbantoglu, associate professor in the department of neurology and neurosurgery at McGill University in Montreal, Canada, received an MDA grant totaling $313,170 for research into increasing levels of the muscle protein utrophin as a therapeutic strategy in Duchenne muscular dystrophy (DMD).
Utrophin is structurally similar to the crucial muscle protein dystrophin, missing in DMD. Nalbantoglu and colleagues aim to validate, in muscle cells and in an animal model of DMD, the potential for utrophin to fulfill some of the functions normally carried out by dystrophin and compensate for the missing protein.
In completed studies, Nalbantoglu's group has shown in an animal model of DMD that utrophin levels can be increased by targeting specific sites in the gene that carries the instructions for utrophin with proteins called transcription factors. Muscle injected with these proteins exhibited fewer hallmarks of disease and increased function despite the absence of dystrophin.
Using the same approach, the team will now refine transcription factors designed to result in increased levels of the human utrophin protein. In parallel, the investigators will explore various methods for delivering the factors throughout the body.
"Funding from MDA is crucial for this work," Nalbantoglu said. The approach of using artificial transcription factors to increase levels of specific genes would have wide application to other muscle diseases. Artificial transcription factors are already in clinical trials for some non-muscle diseases."
Funding for this MDA grant began August 1, 2010.
Grantee: DMD/BMD — Josephine Nalbantoglu
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2011 - DMD/BMD — Melissa Spencer, Ph.D.
Melissa Spencer, professor of neurology and co-director of the Center for Duchenne Muscular Dystrophy at the University of California, Los Angeles, has been awarded an MDA grant totaling $501,493 over three years. The funds will help support Spencer's continuing studies of the role of a protein called osteopontin and of inflammatory processes in a mouse model of Duchenne muscular dystrophy (DMD).
DMD results from a nearly complete absence of the dystrophin protein in the skeletal muscles and heart. Becker muscular dystrophy (BMD), a closely related disease, results from a partially functional dystrophin protein. A large variety of mutations in the dystrophin gene give rise to DMD or BMD.
The osteopontin protein has been shown to play a role in promoting the entry of immune system cells into damaged, dystrophin-deficient muscle, which in turn results in scar-tissue formation (fibrosis).
The Spencer lab has received funding from MDA over the past decade. Spencer and her colleagues, particularly Irina Kramerova and Carrie Miceli, will now study how osteopontin regulates the muscle-invading immune system cells and the manner in which these cells cause fibrosis in mice with a DMD-like disease.
The investigators will use mouse models and cellular and immunological techniques to understand the processes involved. They also will use a specialized mouse that has been genetically engineered to allow for noninvasive imaging of muscles.
These studies will lay the foundation for development of drugs that target osteopontin in DMD or BMD, Spencer said.
"I credit MDA for enabling many of the successes my lab has achieved," she noted.
Funding for this MDA grant began August 1, 2011.
Grantee: DMD/BMD — Melissa Spencer, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - DMD/BMD — Gordon Lynch

MDA awarded $375,000 to Gordon Lynch, professor of physiology at the University of Melbourne, Australia, for research into strategies aimed at improving muscle function in Duchenne muscular dystrophy (DMD).
Lynch's research builds on previous studies conducted by his group and others that have demonstrated the potential of proteins called "growth factors" to preserve muscle fibers. These growth factors are regulated by a family of proteins called insulin-like growth factor binding proteins (IGFBPs).
Lynch's team has uncovered evidence that a particular IGF binding protein called IGFBP-2 may play a key role in the cycles of muscle fiber damage and repair that occur in DMD.
Using a mouse model genetically altered to exhibit either increased or decreased IGFBP-2 activity, the team will seek to determine whether the protein aggravates or improves the DMD disease process. The researchers also will study the effects of IGFBP-2 and other IGF binding proteins on muscle development and muscle repair.
"The MDA has generously supported my laboratory’s research for over a decade, allowing my team to make consistent and significant contributions to the field, including demonstrating the exciting potential of growth factors for improving muscle function in muscular dystrophy," Lynch said. "Support from the MDA has been critical in facilitating my mentorship of some very talented young doctoral students and postdoctoral scientists who have each made important contributions to muscular dystrophy research and become successful independent investigators in their own right."
Lynch's new research will generate important new information about the functional roles of IGF binding proteins in muscular dystrophy, and contribute to the development of therapies designed to improve the quality of life for those with muscle disease.
Funding for this MDA grant began August 1, 2010.
Grantee: DMD/BMD — Gordon Lynch
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2011 - DMD/BMD — Martin Childers, Ph.D.
Martin Childers, a professor at the Institute for Regenerative Medicine in Winston-Salem, N.C., has been awarded an MDA grant totaling $480,000 over three years. The Institute is part of Wake Forest University Baptist Medical Center.
The new funds will help support Childers' research on heart disease associated with Duchenne muscular dystrophy (DMD) and Becker muscular dystrophy (BMD). DMD results from a complete lack of dystrophin protein in skeletal and cardiac muscles, and BMD results from the presence of partially functional dystrophin protein. Both diseases are caused by mutations in the dystrophin gene.
"Many patients with DMD die as young adults from heart failure," Childers said. "The reason for this is that the heart muscle carries a genetic mutation that damages the normal operation of this all-important muscle. Our project will allow for the discovery of new drugs that might reverse or prevent the effects of the disease on the heart muscle."
Childers and colleagues will use two types of new technology to find potential new drugs for heart disease related to dystrophin deficiency. First, they'll use a method called cellular "reprogramming" to create heart cells from the skin cells of DMD patients. "This remarkable new technology will allow us to study how a genetic mutation affects the heart cells of a specific patient," Childers said.
The second method the investigators will employ is a drug-discovery "platform" that can screen thousands of compounds in individual cells.
"By marrying these two incredible technologies, this project will allow, for the first time, the testing of new drugs directly on the heart cells of an individual patient without any risk to the patient," Childers noted.
Funding for this MDA grant began August 1, 2011.
Grantee: DMD/BMD — Martin Childers, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - DMD/BMD — Dawn Cornelison

MDA awarded a grant totaling $160,000 to Dawn Cornelison, assistant professor in the division of biology at the University of Missouri-Columbia, for research into the role of a protein called syndecan-4 in muscle tissue repair after damage.
After muscle injury, Cornelison said, satellite cells (immature muscle cells) move in to make repairs. In order to do their job, the satellite cells need to perform a series of activities: They must first be 'activated' by signals resulting from injury; then divide several times to make more potential muscle cells; then stop dividing; and finally mature, or differentiate, into functional muscle.
In previous MDA-supported work, Cornelison and her research team analyzed a mouse engineered to lack the syndecan-4 protein and found that its satellite cells were unable to perform all of their normal muscle-repair functions. The group found that different parts of the syndecan-4 protein are responsible for regulating different satellite-cell functions in the muscle-repair process.
In its new work the group aims to identify proteins that interact with syndecan-4 and gain a better understanding of how the satellite cells carry out the four activities necessary for muscle repair.
"Funding from MDA has been critical to the progress and development of both this project and my scientific career," Cornelison said. "Particularly in the current environment, with the odds of obtaining funding for general research falling below 10 percent, knowing that MDA is there to sponsor research into muscle disease and regeneration is critical to retaining scientists in the field and encouraging others to undertake research into muscle diseases."
Funding for this MDA grant began August 1, 2010.
Grantee: DMD/BMD — Dawn Cornelison
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2011 - DMD/BMD — Emanuela Gussoni, Ph.D.

MDA has awarded a grant totaling $384,066 over three years to Emanuela Gussoni, an assistant professor in the division of genetics and program in genomics at Children's Hospital Boston and Harvard University. The grant will support Gussoni's studies to improve the efficacy of stem cell transplantation as a strategy to treat Duchenne muscular dystrophy (DMD), Becker muscular dystrophy (BMD) and possibly other forms of muscular dystrophy.
The direct injection of normal muscle stem cells into mice and people with muscular dystrophy has been attempted as a way to provide missing proteins to muscle tissue. (In DMD, the dystrophin protein is missing; in BMD, it's present but only partially functional.)
However, Gussoni said, studies have shown that, while the technique is safe in patients, their muscles have not shown significant improvement. One of the causes for the lack of improvement is the rapid death of the normal cells following transplantation.
To overcome this problem, different cell types more capable of surviving the transplant and producing the desired proteins must be identified. Gussoni's research team has found that human cells that produce a surface protein called MCAM can form muscle in tissue culture and following injection into animals.
If they live up to the preliminary expectations of Gussoni and her colleagues, MCAM-expressing cells could become candidates for cell transplantation to treat MD.
"MDA funding has been instrumental throughout my career," said Gussoni, who has received several MDA grants. "MDA develops and fosters both new and established investigators who are highly committed to improving the lives of people with muscle disorders."
Funding for this MDA grant began August 1, 2011.
Grantee: DMD/BMD — Emanuela Gussoni, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - DMD/BMD — Carmen Bertoni
MDA awarded $408,915 to Carmen Bertoni, assistant professor of neurology at the University of California, Los Angeles, to continue research into DNA repair strategies in Duchenne muscular dystrophy (DMD).
Bertoni's research group has pioneered a technology that uses small molecules called single-stranded oligonucleotides, or ssODNs, to act directly on the source of the problem in DMD: the "genetic blueprint," or DNA. In people with DMD, the DNA that makes up the dystrophin gene contains errors that lead to missing or nonfunctional dystrophin protein.
"We use ssODNs to let the muscle know of those errors and give the opportunity to each cell that composes muscles to correct the mistake," Bertoni said. The fix is permanent, negating the need for ongoing treatment.
The process involves injection of the oligonucleotide into muscle, where it seeks the flawed region of DNA and highlights the specific error. The cell is alerted to the presence of the genetic defect, and activates the repair process to correct the flaw.
To date, the main limitation of ssODN-mediated gene correction has been the low frequency of gene repair. Bertoni's group aims to increase the strategy's efficiency to generate levels of gene repair that are clinically meaningful as a viable treatment for DMD.
The group will test the oligonucleotides first in muscle cells and then, once investigators have optimized the structure, in dystrophin deficient mice.
"MDA has had and continues to have a pivotal role in the development of this technology for the treatment of DMD," Bertoni said. "Without any question, the advance of clinical applications to DMD using ssODNs heavily relies on the help that continues to be received by the MDA. It is a great honor to be part of the numerous and extremely talented scientists that are supported by the Association."
Funding for this MDA grant began August 1, 2010.
Grantee: DMD/BMD — Carmen Bertoni
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - DMD/BMD — Bernard Jasmin

MDA awarded Professor Bernard Jasmin, vice-dean of research at the University of Ottawa in Canada, a grant totaling $360,000 to study the impact of small molecules called "exercise mimetics" in Duchenne muscular dystrophy (DMD).
One approach to counteract the effects of the loss of the muscle protein dystrophin in DMD involves increasing levels of a similar structural protein called utrophin. Studies performed in a widely used mouse model of DMD, the mdx mouse, have shown that increased utrophin expression can compensate for the lack of dystrophin and ameliorate symptoms of the disease.
Because utrophin activity normally is low and focused only in certain regions of the muscle, Jasmin's laboratory looks for ways to increase levels of the protein in DMD muscles.
"Our first preclinical study showed that treatment of mdx mice with an exercise mimetic known as GW501516 resulted in an increase in utrophin expression, improved muscle function and improved pathological symptoms of the disease," Jasmin said.
His current work builds on the previous study results, with plans for continued testing of GW501516 in the mdx mouse model of DMD, and expansion of the research to include testing of other molecules including AICAR, beta-GPA and resveratrol.
Because of the possible effects of exercise mimetics on utrophin in DMD-affected muscles, the small molecules hold potential for dramatic improvement in quality of life for individuals with the disease. GW501516 currently is under development to treat obesity by the pharmaceutical company GlaxoSmithKline, which could help speed its development for therapeutic use in people with DMD.
"MDA funding has allowed us to take a relatively basic molecular observation and perform research that eventually could result in development of novel therapeutic approaches for treatment of people with DMD," Jasmin said.
Funding for this MDA grant began August 1, 2010.
Grantee: DMD/BMD — Bernard Jasmin
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2011 - DMD/BMD — Alessandra Sacco, Ph.D.

MDA has awarded a research grant totaling $447,092 over three years to Alessandra Sacco, an assistant professor in the Muscle Development and Regeneration Program at Sanford-Burnham Medical Research Institute in La Jolla, Calif. The grant will help support Sacco's research on early-stage transplantation of muscle stem cells to treat a mouse model of Duchenne muscular dystrophy (DMD).
The goal of Sacco's new study is to understand whether transplanting muscle stem cells into a DMD mouse model during the fetal or newborn period can prevent the muscle degenerative process seen in DMD.
Sacco said that, by the time DMD is diagnosed and treatment begins in children with the disease, significant damage to muscles has accumulated, which "poses a major hurdle to the design of efficient therapeutic approaches."
She said transplantation of muscle stem cells during fetal or neonatal life may enhance the regenerative potential of skeletal muscles, resulting in an improved therapeutic outcome.
Sacco's group will make use of a recently developed DMD mouse model, in which the mice lack two proteins. Like people with DMD, they lack dystrophin, a muscle protein that's completely absent in this disease and only partially functional in people with the related Becker muscular dystrophy (BMD); and they also lack telomerase, a repair enzyme. These mice have a severe DMD-like disease.
If the transplantation approach is successful in these mice, it could have applications to human DMD, BMD and perhaps other forms of MD.
"MDA has been fundamental to my career development," Sacco said. "During my postdoctoral training, I received an MDA development grant for studying the contribution of bone-marrow-derived cells to skeletal-muscle regeneration. In the difficult funding climate of the last few years, the efforts of this organization to promote research in this field have been outstanding."
Funding for this MDA grant began August 1, 2011.
Grantee: DMD/BMD — Alessandra Sacco, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - DMD/BMD — Ashok Kumar

MDA awarded associate professor Ashok Kumar at the University of Louisville School of Medicine in Louisville, Ky., a grant totaling $349,206 for continued study of the molecular mechanisms that underlie disease onset and progression in Duchenne muscular dystrophy (DMD).
Kumar's team will work to identify biochemical changes in skeletal muscle caused by the loss of functional dystrophin protein in the disease.
"Recent studies in my laboratory have identified a set of proteins known as 'matrix metalloproteinase' (MMPs) that are highly deregulated in dystrophic muscles," Kumar said. "Interestingly, MMPs are the major enzymes responsible for tissue degradation in a number of disease states and conditions."
Kumar and colleagues have found that treatment with a molecule called batimastat is highly effective at ameliorating disease symptoms such as fiber necrosis (death) and inflammation, and improving skeletal muscle structure in dystrophin-deficient mice.
Now, the team will investigate the therapeutic potential of MMP-inhibiting drugs, some of which are already being used for other diseases in humans (including chemical relatives of the antibiotic tetracycline).
"I strongly believe that the current funding provided by MDA will open new avenues for treatment of DMD in the near future," Kumar said. "I sincerely thank MDA for its generous funding to pursue my research work in DMD."
Funding for this MDA grant began August 1, 2010.
Grantee: DMD/BMD — Ashok Kumar
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - CMT — Albena Jordanova

MDA awarded a grant totaling $282,630 to Albena Jordanova, professor in the department of genetics at the University of Antwerp, Belgium, for research into the molecular causes of, and potential drug targets for, a recently discovered form of Charcot-Marie-Tooth (CMT) disease known as dominant intermediate CMT type C (DI-CMTC).
Jordanova's research team was the first to describe the new CMT type and showed that the disease is caused by different mutations (genetic flaws) in the gene that carries instructions for an enzyme called tyrosyl-tRNA synthetase (YARS).
In their new work, Jordanova and colleagues aim to identify genes that modify the neurodegenerative symptoms of DI-CMTC present in a fruit fly research model of the disease. The team will then test the modifying genes to determine how well each is able to interact with drug-like chemical compounds, in an effort to pinpoint the best drug targets for DI-CMT and other types of the disease.
The "best gene hits" will be those that carry instructions for proteins that are both structurally favorable — that is, their protein folds are amenable to interactions with drug-like chemical components — and functionally favorable with a proven ability to affect the DI-CMTC disease process.
"MDA funding is absolutely essential to start this work," Jordanova said. "This is important seeding money for us to initiate a midterm research program and to perform the research we dream of."
Funding for this MDA grant began August 1, 2010.
Grantee: CMT — Albena Jordanova
Grant type: Research Grant
Award total:
Institution:
Country:
Grant – Winter 2011 – DMD - David Goldhamer, Ph.D.

MDA has awarded a research grant totaling $375,000 over three years to David Goldhamer, associate professor, director of the Center for Regenerative Biology, and associate director of the UConn Stem Cell Institute at the University of Connecticut in Storrs. The new funds will help support Goldhamer’s study of muscle stem cells and the repair of damaged muscle in Duchenne muscular dystrophy (DMD).
"Satellite" cells are adult muscle stem cells that are responsible for postnatal muscle growth and the repair of damaged muscle in injury and disease. In DMD, accumulation of intramuscular fat and connective tissue represent hallmarks of advanced disease, and the presence of these non-muscle tissues significantly affects muscle structure and function.
Satellite cells have been implicated as a possible cell of origin for increased fat and fibrotic (scarred) tissue in DMD muscle, but their involvement remains uncertain and controversial.
Using a research mouse model with a DMD-like disease, Goldhamer and colleagues will examine whether satellite cells promote increased fat and fibrosis in diseased muscle. In addition, the team will study regulators of muscle regeneration that may interact with or regulate satellite cell functions.
Results from Goldhamer’s work may help determine the potential benefits of modulating satellite cell behavior as a therapeutic strategy in DMD.
Funding for this MDA grant began February 1, 2011.
Grantee: DMD - David Goldhamer, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2011 - DMD — Margaret Zacharin, M.D.

Margaret Zacharin, an associate professor in the department of endocrinology and diabetes at Royal Children's Hospital in Parkville, Victoria, Australia, has been awarded an MDA research grant totaling $268,021 over two years. The award will help support a clinical trial of the drug zoledronic acid in children and adolescents with Duchenne muscular dystrophy (DMD).
Zoledronic acid is part of a class of medications known as bisphosphonates, which are used to prevent bone loss in osteoporosis and other bone conditions.
Children with DMD lose bone in part because of their underlying disease and in part because of the effects of corticosteroid medications. These are commonly used to prolong muscle function in DMD but have adverse effects on bone density.
"Bisphosphonates alter the course of corticosteroid-induced bone loss and largely prevent this complication in the adult population," Zacharin said. "It's more difficult to provide such evidence in a pediatric population." However, she noted, with newer techniques of calculation available, more accurate data can now be obtained.
"We aim to determine whether intravenous zoledronic acid plus vitamin D and calcium is better than vitamin D and calcium alone in increasing spinal bone density, reducing bone turnover and/or reducing fracture rate over two years in boys with Duchenne muscular dystrophy."
Zacharin will work closely with Craig Munns, an associate professor in the Department of Endocrinology at Children’s Hospital in Westmead, New South Wales, Australia, on this project.
She said results of the zoledronic-acid study may be used to develop a larger trial to assess fracture reduction in the DMD population and might ultimately be extended to other diseases, including other neuromuscular disorders.
MDA support, she said, "will allow us to undertake and complete this important study in a timely fashion."
Funding for this MDA grant began August 1, 2011.
Grantee: DMD — Margaret Zacharin, M.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - CMS — Michael Francis

Michael Francis, associate professor in the department of neurobiology at the University of Massachusetts Medical School in Worcester, received an MDA grant totaling $330,000 for research into effects on the connection between nerve and muscle known as the neuromuscular junction, or NMJ, in congenital myasthenic syndrome (CMS).
Francis and colleagues have engineered a research model using C. elegans, a type of worm also known as a nematode, that mimics many of the key features of a form of CMS called slow-channel congenital myasthenic syndrome (SCCMS). The team will use sophisticated genetic tools paired with measurements of the electrical properties such as voltage changes and electric currents in cells and tissues, and a sophisticated imaging technique called confocal microscopy in its analysis of NMJ function in both normal and diseased tissues.
The investigators plan also to pinpoint the molecular mechanisms underlying degeneration and alterations in NMJ function caused by neuromuscular disorders.
"We expect our efforts will provide key mechanistic insights into conserved molecular pathways important for NMJ development and function in the normal organism, as well as during diseases that impact NMJ function, such as SCCMS," Francis said. "Identifying the components of these pathways will be important for the development of new therapeutic strategies to combat neuromuscular disorders."
Funding for this MDA grant began August 1, 2010.
Grantee: CMS — Michael Francis
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2011 - DMD — Jasprina Noordermeer, Ph.D.

MDA has awarded a research grant totaling $278,570 over three years to Jasprina Noordermeer, a professor in the department of molecular cell biology at Leiden University Medical Center in the Netherlands. The grant will support Noordermeer's studies of the role of dystrophin in the brain. The dystrophin protein is absent or deficient in boys with Duchenne muscular dystrophy (DMD).
The role of dystrophin in skeletal muscles is fairly well-understood, and its absence in DMD leads to the progressive weakness seen in this disease. A great deal is also known about the role of this protein in cardiac muscle, where its absence leads to cardiac muscle abnormalities (cardiomyopathy) in DMD.
It's also known that dystrophin is normally present in the brain, and that at least some DMD patients have cognitive disabilities. However, little is known about the role of dystrophin or the effects of dystrophin deficiency in the brain.
Ultimately, Noordermeer says, treatments for DMD should reverse both muscle loss and the cognitive deficits associated with this disease, but how the lack of dystrophin results in defects at the connections ("synapses") where nerve cells interact with each other is unclear.
She and her colleagues will study the role of dystrophin and dystrophin deficiency in the brain in dystrophin-deficient flies and mice.
"MDA funding is critical to allow us to further our understanding of the functions of dystrophin and its [molecular] partners at the synapse," Noordermeer said. "We particularly appreciate MDA's commitment to funding research in model organisms directed toward unraveling DMD-associated dysfunctions."
Funding for this MDA grant began August 1, 2011.
Grantee: DMD — Jasprina Noordermeer, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - CCD — Francesco Muntoni
MDA awarded a grant totaling $375,000 to Francesco Muntoni, professor of pediatric neurology at University College London (UCL), United Kingdom, for research into the molecular mechanisms underlying central core disease (CCD) and multiminicore myopathies. Muntoni and Michael Duchen, professor of physiology, and cell and developmental biology, also at UCL, will work together, focusing on the mechanisms that lead to muscle weakness in the two diseases.
CCD and multiminicore myopathies both are caused by mutations in the ryanodine receptor gene (RYR1), which carries instructions for the RYR1 protein.
"While we have known for a number of years that RYR1 gene mutations were associated with these core myopathies, the precise mechanism leading to muscle weakness in affected individuals is less clear," Muntoni said.
It's known that RYR1 plays a key role in the release of calcium in skeletal muscle; the calcium release is followed by muscle contraction.
Muntoni and Duchen will investigate the hypothesis that chronic calcium dysregulation due to dysfunctional RYR1 protein leads to malfunction, in each muscle fiber, of the cell's energy factories, called mitochondria. With muscle cells from individuals carrying RYR1 mutations, the two will study various aspects of mitochondria health and function following exposure to abnormal calcium release.
"This study is a follow-on of a currently funded MDA grant that is coming to an end," Muntoni said. "The current MDA grant allowed us to improve our insight on the correlation between different mutations in RYR1 and the core myopathies. Without the original MDA grant, we would not have been in a position to establish these novel correlations between RYR1 and congenital diseases."
An improved understanding of the link between dysregulated calcium handling and mitochondria dysfunction could lead to new therapeutic interventions for individuals with core myopathies.
Funding for this MDA grant began August 1, 2010.
Grantee: CCD — Francesco Muntoni
Grant type: Research Grant
Award total:
Institution:
Country:
Grant – Winter 2011 – DMD - Bradley Olwin, Ph.D.
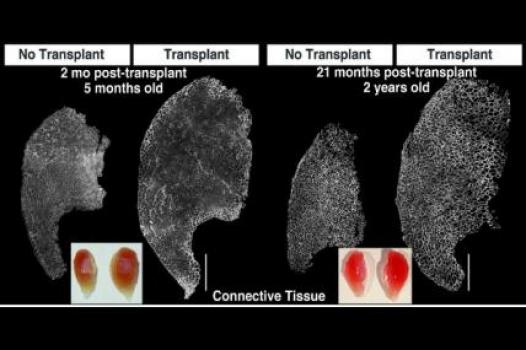
MDA has awarded a research grant totaling $369,165 over three years to Bradley Olwin, professor of molecular, cellular & developmental biology at the University of Colorado in Boulder. The new funds will help support Olwin’s study of muscle regeneration in injured and diseased skeletal muscle — particularly in the muscular dystrophies, including Duchenne muscular dystrophy (DMD).
Olwin is a longtime MDA grantee, having received funding from the Association almost continuously since the early 1980s.
Diseases such as the muscular dystrophies that result in progressive loss of skeletal muscle tissue reflect disruption of the normal muscle regenerative processes. If the cells responsible for normal repair could be augmented by drug therapies or cell replacement therapies, regeneration might be enhanced, slowing or halting the loss of skeletal muscle function.
In previous studies, Olwin and colleagues identified a rare stem cell that they hypothesize is a primary source of skeletal muscle stem cells (also called "satellite" cells). The team plans to study the newly identified cells and uncover any contributions they may make to the regeneration process in injured and diseased skeletal muscle. Next, the team will transplant the cells into the muscles of a research mouse with a DMD-like disease in order to determine how well they restore muscle function, slow muscle scarring (called fibrosis), and produce the protein missing in DMD, dystrophin.
Findings from Olwin’s studies may result in new drug-based or cell-based strategies to enhance new muscle growth in progressive muscle diseases.
"I have been funded by the MDA for the majority of my career as a research scientist, beginning in the early 1980s," Olwin said. "Without MDA funding, I may not have continued in skeletal muscle research."
Funding for this MDA grant began February 1, 2011.
Grantee: DMD - Bradley Olwin, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2011 - DMD — Diego Fraidenraich, Ph.D.

Diego Fraidenraich, an assistant professor in the department of cell biology & molecular medicine at the University of Medicine and Dentistry of New Jersey, was awarded an MDA grant totaling $375,000 over three years. The funds will support his study of the relationship between muscle and fat formation in a mouse model of Duchenne muscular dystrophy (DMD).
These mice, like humans with DMD, lack the dystrophin protein in their skeletal muscles and heart because of a mutation in the dystrophin gene. They develop a muscle disease resembling human DMD.
Recently published reports have shown that skeletal muscle and fat have a common cellular ancestor, and Fraidenraich's team intends to understand the similarities and differences in the development of these two tissues.
In previous experiments, Fraidenraich and colleagues treated DMD mice with stem cells from healthy mice, so that the dystrophin protein was present in some, but not all, of their cells.
These so-called "mosaic" mice developed some normal muscle tissue from the stem cells, as well as some fat tissue that had muscle-like characteristics. The researchers termed this unusual, stem-cell-derived tissue "muscularized fat."
In his new work, Fraidenraich will seek to understand more about this muscle-like fat derived from the transplanted stem cells and define its possible role in the development of muscle. He'll also try to increase the ratio of muscle to fat in the DMD mice.
Fraidenraich hopes to provide new understanding of muscle formation that will supply leads for future therapies for DMD and perhaps other forms of muscular dystrophy.
He has received MDA funding since 2007, and says, "Continuous funding from MDA has allowed me to develop and expand my research program. The support has been instrumental to my research and my lab."
Funding for this MDA grant began August 1, 2011.
Grantee: DMD — Diego Fraidenraich, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - ALS — Wilfried Rossoll

MDA awarded $358,653 to Wilfried Rossoll, assistant professor at Emory University in Atlanta, for research into the effects on nerve cells, or "motor neurons," of toxic TDP43 protein, implicated in ALS (amyotrophic lateral sclerosis, or Lou Gehrig's disease).
"Recently, we have developed novel tools to study protein localization, trafficking and function in cultured primary motor neurons," Rossoll said. "Funding from MDA will now allow us to apply these methods to address the biological role of the ALS disease protein TDP43 in motor neurons."
TDP43 protein is present in all body tissues and serves multiple roles in the processing of messenger RNAs (mRNAs), the molecules that carry genetic information from DNA to the sites of protein synthesis. However, the function of TDP43 in motor neurons and vulnerability of those cells to the protein still is not understood.
"Most studies on the role of TDP43 protein have used non-neuronal cell lines, so its function in the highly specialized motor neuron cell type remains unclear," Rossoll said. "We will use cell cultures of primary motor neurons and motor neurons generated from stem cells to study the function of TDP43 in this cell type, and to gain information about its involvement in ALS."
Preliminary data generated by Rossoll's team suggests that TDP43 plays a role in mRNA processing in the long axon fibers that conduct electrical impulses to muscle cells. The team will test the hypothesis that decreased levels of TDP43 in mRNA-protein complexes in these axons may contribute to the degeneration of motor neurons in ALS.
Increased understanding of the molecular and cellular functions of TDP43 could help lead to the development of therapies that delay or even prevent motor neuron degeneration in people with ALS or other neurodegenerative diseases.
Funding for this MDA grant began August 1, 2010.
Grantee: ALS — Wilfried Rossoll
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - ALS — Stanley H. Appel

MDA awarded a research grant totaling $330,000 to Stanley H. Appel, chair of the department of neurology at the Methodist Neurological Institute (MNI) in Houston, to study the protective effects of a specific class of immune system cells in ALS (amyotrophic lateral sclerosis, or Lou Gehrig's disease).
Appel, a longtime MDA adviser and director of the MDA/ALS center at MNI, will look at T cells, observed in people with ALS as well as in mouse models of the disease, and known to be involved in protecting motor neurons (nerve cells that activate muscles).
Appel's lab previously has documented that T cells can protect neurons in a mouse model of ALS, at least in part by enhancing the neuroprotective functions of another type of immune system cell called microglia. When activated, these cells can produce helpful proteins, but they also can cause dangerous inflammation.
Finding ways to protect motor neurons is a key goal in the development of ALS therapeutics, and Appel's group is working to understand the interaction among other cells in the immune system responsible for prompting T cells to protect neurons.
Project plans include the transplantation of various T-cell types into ALS research mouse models in order to determine which of the cells are most helpful to neurons. A greater understanding of this T-cell population and its associated molecular signals may lead to therapeutics based on increasing the numbers and effectiveness of these cells to help protect neurons in people with ALS.
"MDA has been a critical source of support throughout the development of our immunological studies in ALS," Appel said. "It's permitted us to assemble a team of scientists focused on the neuroinflammatory processes in ALS, and to substantiate the importance of protective immunomodulation in ALS."
Funding for this MDA grant began August 1, 2010.
Grantee: ALS — Stanley H. Appel
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - ALS — Shanthini Sockanathan
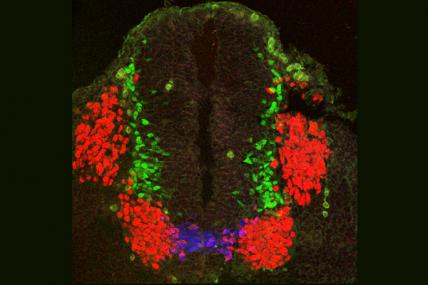
MDA awarded a grant totaling $347,832 to Shanthini Sockanathan, associate professor of neuroscience at the Johns Hopkins University School of Medicine in Baltimore, for research into the molecular causes of nerve cell, or motor neuron, degeneration in diseases including ALS (amyotrophic lateral sclerosis, or Lou Gehrig's disease).
Sockanathan and colleagues will study the Gde2 mouse, a new model of motor neuron degeneration that they developed based on studies of the maturation, or "differentiation," of motor neurons in embryonic mouse spinal cords.
The Gde2 mouse lacks a protein called GDE2. Mouse embryos lacking the protein possess significantly decreased numbers of motor neurons, but at birth they contain numbers of these cells equivalent to those of control mice. As adults, the Gde2 mice experience a significant loss of motor neurons, with the remaining nerve cells indicating progressive degeneration.
In their new work, the investigators will study the physical and biological characteristics of Gde2 mice, and monitor the progression of motor neuron degeneration in older animals. Because the GDE2 protein normally is active in both differentiating and fully matured motor neurons, the team plans to remove it in mice at different times during development to determine at which stage or stages GDE2 loss causes degeneration.
"MDA's clear commitment to funding basic research projects has been instrumental in my ability to develop and maintain an active research program dedicated to understanding the mechanisms of neuronal differentiation," Sockanathan said. "Indeed, my first grant as an independent investigator was from MDA, and this funding helped me establish my research program and, importantly, provided me with the flexibility to chart a new direction in my work. Over the years, I have been fortunate to receive more funding from MDA, and in each case this funding has helped me broaden my research program in novel areas."
Funding for this MDA grant began August 1, 2010.
Grantee: ALS — Shanthini Sockanathan
Grant type: Research Grant
Award total:
Institution:
Country:
Grant – Winter 2011 – CNM/MTM - Jocelyn Laporte, Ph.D.
MDA has awarded a research grant totaling $341,250 over three years to Jocelyn Laporte at the University of Strasbourg, France. The new funds will help support Laporte’s efforts to identify genes responsible for centronuclear/myotubular myopathies (CNM)/(MTM).
Laporte noted that his lab identified some of the first genes associated with these types of diseases in 1996, but that a number of other CNM-causing genes remain to be identified. Using a sophisticated technology called "high-throughput sequencing," also known as "massively parallel sequencing," Laporte and colleagues plan to uncover new genes associated with CNM.
The main challenge of this novel approach is the identification of the causative mutation among the many DNA variants that are not connected to the myopathy. It will require special effort to analyze the data.
Findings from Laporte’s work likely will facilitate diagnosis of CNM in individuals, improve genetic counseling services, and lend insight to better care and follow-up after diagnosis. In addition, the identification of causative genes will help inform scientists of the underlying disease mechanisms, help researchers determine eligibility for human clinical testing, and provide new targets at which to aim potential new therapies.
"MDA funding is a key factor for successful research on rare muscle diseases," Laporte said.
Funding for this MDA grant began February 1, 2011.
Grantee: CNM/MTM - Jocelyn Laporte, Ph.D.
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2010 - ALS — Oliver Hobert
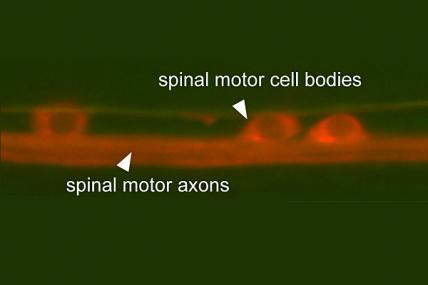
MDA awarded $374,511 to Oliver Hobert, professor of biochemistry and molecular biophysics at Columbia University, New York, to study the function of the TDP43 gene, mutations in which can cause ALS (amyotrophic lateral sclerosis, or Lou Gehrig's disease).
"In order to diagnose and treat ALS, it is important to understand the molecular events that underlie this disease," Hobert said. The TDP43 gene, known to cause ALS in humans, "works in a manner that is not understood. Our goal is to better understand the function of this gene."
Using the invertebrate C. elegans (nematode, or roundworm) model, Hobert's team will study the TDP43 gene in order to determine its function and possible interaction with other genes.
"Funding by the MDA means a great deal to us — not just because of the financial support but also because it motivates us to direct our basic research toward questions that are medically relevant," Hobert said.
Funding for this MDA grant began August 1, 2010.
Grantee: ALS — Oliver Hobert
Grant type: Research Grant
Award total:
Institution:
Country:
Grant - Summer 2011 - CMD — Sonja Nowotschin, Ph.D.

Sonja Nowotschin, a postdoctoral research fellow in the developmental biology department at the Sloan-Kettering Institute in New York, has been awarded an MDA development grant totaling $163,638 over three years. (Development grants are MDA's mechanism for furthering the career development of promising young researchers.)
The funding will help support Nowotschin's research on how muscles are formed at the embryo stage, with the goal of understanding and ultimately treating congenital muscular dystrophies (CMDs) and congenital myopathies, such as myotonia congenita, paramyotonia congenita,central core disease (CCD), nemaline myopathy,myotubular/centronuclear myopathies (MTM/CNMs) and periodic paralysis.
Nowotschin and her colleagues will study embryonic mice to investigate the origin and development of the skeletal muscles. They'll identify and study the stem cells that form muscles in mice with and without genetic mutations.
"Knowledge gained from these studies will assist logical efforts to design new therapeutic strategies," Nowotschin said.
"Importantly," she added, "by investigating the molecular mechanisms operating in the embryo, we will be able to formulate the molecular principles that should assist in developing methods to reprogram adult, or 'differentiated,' cells." Cells that have matured (differentiated) into muscle tissue and have then been reprogrammed back into stem cells are being considered as possible therapies to treat muscle disease.
MDA funding, Nowotschin said, "is essential to move this project forward."
Funding for this MDA grant began August 1, 2011.
Grantee: CMD — Sonja Nowotschin, Ph.D.
Grant type:
Award total:
Institution:
Country:
Learn more about the research projects MDA is currently funding:
- MDA Awards 25 Grants Totaling More Than $6.6 Million for Neuromuscular Disease Research
- Muscular Dystrophy Association Awards 26 Grants Totaling More Than $7.5 Million for Neuromuscular Disease Research
Research Across Diseases
As part of MDA's basic research program, the grants we fund focus on advancing basic science and generating ideas for potential drug therapies through projects initiated by the researchers themselves. Through the projects they fund we will learn more about the processes that drive neuromuscular diseases. We’ll identify, validate and optimize biological targets at which to aim future therapies. We’ll test potential therapeutic strategies, develop drug development tools and make other advances that will help pave the way to more clinical trials.
Twice a year, grant applications are reviewed by MDA’s Research Advisory Committee which recommends the best projects for approval. Funding is approved by MDA’s Board of Directors.
- MDA Medical Education
- Grants at a Glance
- Research Grants
- Creating a New Therapy
- What We've Achieved
- MDA Venture Philanthropy
- MDA Annual Conference
- MOVR Data Hub
- Newborn Screening for Neuromuscular Diseases
- Cost of Illness of Neuromuscular Diseases in the US
- Contact Our Research Team
- MDA Kickstart Program
- Telemedicine Resources
Find MDA
in your Community
-

Search for Clinical Trials
Learn More -

Grants at a Glance
Read More